Role of NRF2 in the Pulmonary Response to Inhaled Mine Tailing Dust
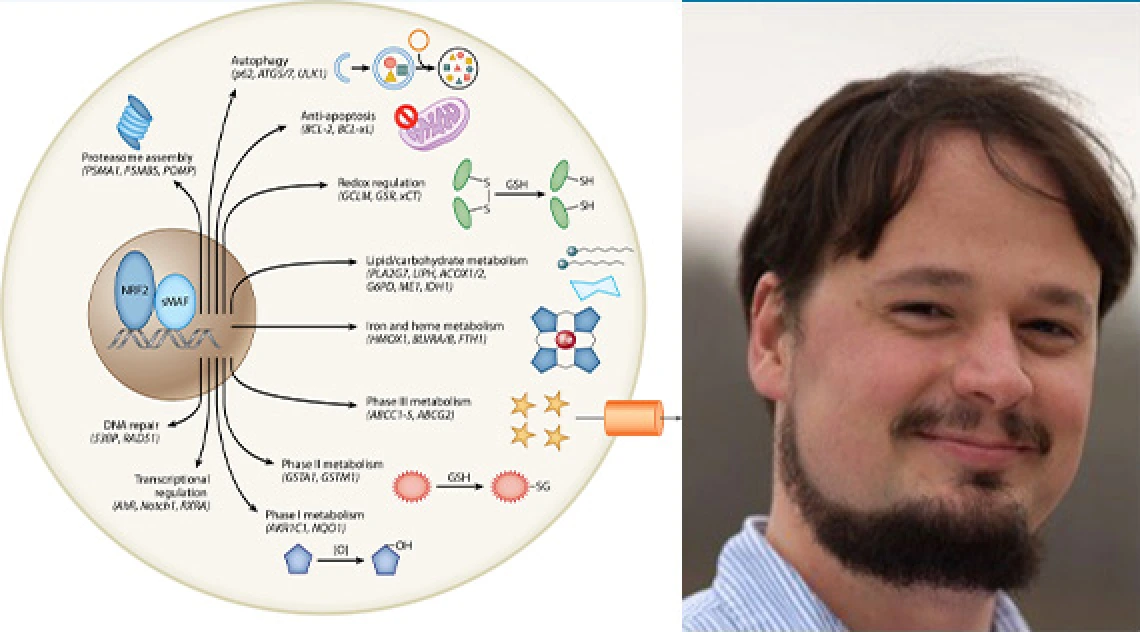
 Chronic environmental exposure to unsafe levels of arsenic has been linked to adverse health outcomes, including lung disease. The mechanisms underlying arsenic dust toxicity in lung tissue have not been fully elucidated. We have recently demonstrated that arsenic exposure activates the Nrf2 pathway and that while acute activation of Nrf2 can be protective, prolonged activation of Nrf2, which may occur in populations chronically exposed to arsenic, may be harmful. We have worked to elucidate the mechanism by which arsenic activates Nrf2 (i.e. acute vs. prolonged), and to determine if acute activation of Nrf2 prevents arsenic dust-induced lung injury. Our work shows that environmentally relevant levels of arsenic do not increase oxidative stress, but mainly disrupt normal cellular proteostasis, challenging the currently accepted notion in the field regarding how arsenic promotes disease progression. We have made critical discoveries regarding how arsenic exposure leads to lung injury, including the mechanism by which arsenic blocks autophagy, a key pathway whose dysfunction leads to chronic Nrf2 activation, and that rescuing this blockage restores normal Nrf2 signaling. Taken together, these findings indicate that compromised autophagy, and not enhanced oxidative stress occurs early during arsenite exposure, and that restoring the autophagy pathway and proper proteostasis could be a viable option for treating arsenic-linked diseases.
Chronic environmental exposure to unsafe levels of arsenic has been linked to adverse health outcomes, including lung disease. The mechanisms underlying arsenic dust toxicity in lung tissue have not been fully elucidated. We have recently demonstrated that arsenic exposure activates the Nrf2 pathway and that while acute activation of Nrf2 can be protective, prolonged activation of Nrf2, which may occur in populations chronically exposed to arsenic, may be harmful. We have worked to elucidate the mechanism by which arsenic activates Nrf2 (i.e. acute vs. prolonged), and to determine if acute activation of Nrf2 prevents arsenic dust-induced lung injury. Our work shows that environmentally relevant levels of arsenic do not increase oxidative stress, but mainly disrupt normal cellular proteostasis, challenging the currently accepted notion in the field regarding how arsenic promotes disease progression. We have made critical discoveries regarding how arsenic exposure leads to lung injury, including the mechanism by which arsenic blocks autophagy, a key pathway whose dysfunction leads to chronic Nrf2 activation, and that rescuing this blockage restores normal Nrf2 signaling. Taken together, these findings indicate that compromised autophagy, and not enhanced oxidative stress occurs early during arsenite exposure, and that restoring the autophagy pathway and proper proteostasis could be a viable option for treating arsenic-linked diseases.
Publications
Dodson, M., M. Rojo de la Vega, B. Harder, R. Castro-Portuguez, S.D. Rodrigues, P.K. Wong, E. Chapman, D.D. Zhang. 2018. Low-level arsenic causes proteotoxic stress and not oxidative stress. Toxicol. Appl. Pharmacol. 341:106-113.
Dodson, M., M. Rojo de la Vega, A.B. Cholanians, C.J. Schmidlin, E. Chapman, and D.D. Zhang. 2018. Modulating NRF2 in disease: timing is everything. Annual Rev. Pharmacol. Toxicol. doi: 10.1146/annurev-pharmtox-010818-021856.

